In competitive areas of biomedical research, such as drug discovery and vaccine development, choosing the right technique for your research needs could save you valuable time and resources. For Instance, ELISAs can confirm the binding of a large number of interactions. This makes this technique an ideal choice for high-throughput drug target screening.1 However, ELISAs provide limited kinetics data about interactions beyond steady-state (e.g. endpoint) affinity values. Thus, an ELISA would not be a viable option if detailed kinetic rate information about an interaction is needed.1
Choosing the right technique for your research holds significant value in both time and resources. At Nicoya, we specialize in surface plasmon resonance (SPR), having developed the OpenSPRTM to provide accessible SPR instrumentation for researchers. A technique rapidly growing in popularity right now is surface plasmon resonance imaging (SPRi), a higher throughput SPR technique. In a drug discovery workflow, SPRi is frequently considered for obtaining binding kinetics data of a large number of drug candidates. However, the sensitivity limitations of SPRi may not always meet the intense criteria in drug and vaccine development.
So when is it best to use surface plasmon resonance imaging, and when does SPRi fall short of your research needs? In this blog post, we elaborate on the main differences between the optical system used in SPRi and those used in other SPR techniques. Through these differences, we highlight where each technique fits into your research workflow.
Traditional Surface Plasmon Resonance
To sufficiently describe surface plasmon resonance imaging, the concept of traditional SPR should first be reviewed. It should be noted that the OpenSPRTM uses localized surface plasmon resonance (LSPR), a principle that is covered below.
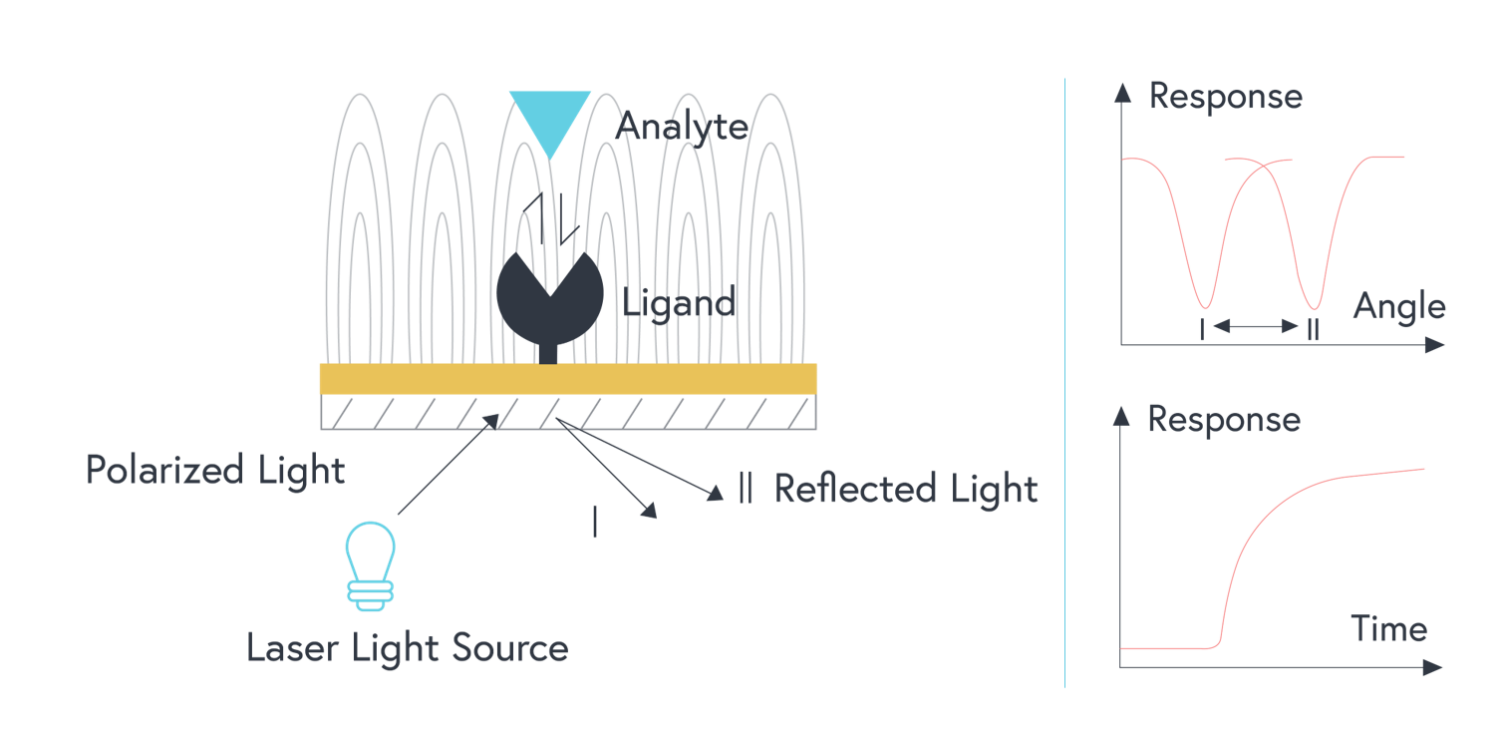
In a traditional SPR system, polychromatic light is shone through a prism onto the thin film of gold.2,3 When the light hits the metal film at a particular angle (the angle of incidence), a portion of the light energy excites electrons on the metal surface.2,3 This causes them to oscillate and generate an electric field – this is the phenomenon known as surface plasmon resonance.2
The angle of incidence is strongly affected by the refractive index of material bound near the gold surface.2 When molecules are bound to the metal surface, the refractive index increases, thus changing the angle of incidence.2 As analytes bind to interacting partners that have been immobilized to the gold surface, the change in angle of incidence is measured to produce a response curve in real-time.2 From this, a wealth of information about the binding interaction can be extrapolated – including the on rate, off rate, and overall affinity of the interaction.
This direct, real-time method of measurement is very sensitive but is limited to a small number of channels.3 The sensitivity of any SPR system using a film of gold is also limited by a larger field decay length compared to that of localized surface plasmon resonance (covered below). As a result, measurements on traditional SPR systems are more sensitive to noise, such as bulk shifts.
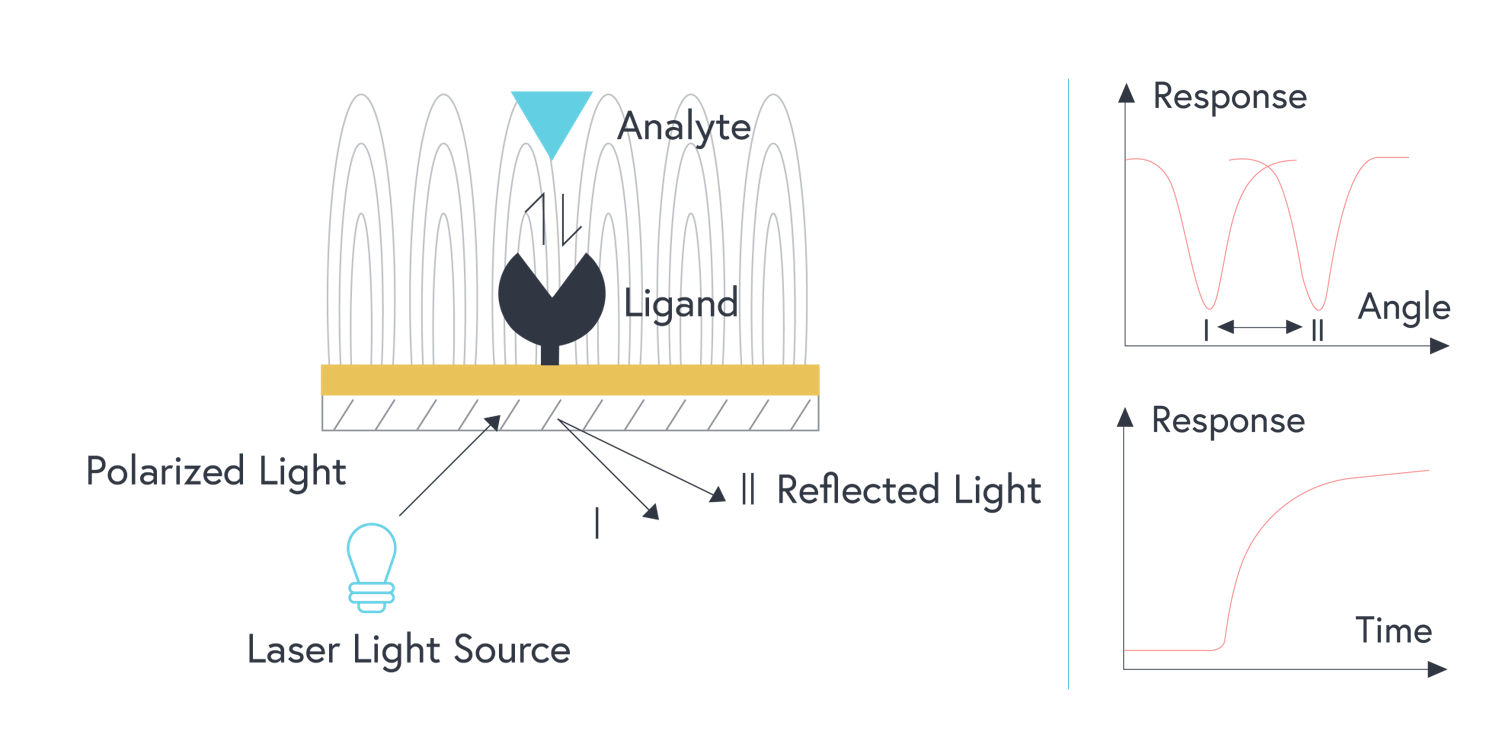
Figure 1: Illustration of traditional SPR optical system (prism is not shown). Polarized light shone onto a film of gold with a ligand immobilized to the surface. As analytes bind to the surface, the resulting change in the refractive index changes the angle of incidence that is measured.
What is Surface Plasmon Resonance Imaging (SPRi)?
Also known as surface plasmon resonance microscopy, SPRi follows the same general principles of SPR. However, the information that is measured and the method of detection is different. This technique was inspired by the need for a higher-throughput method of studying binding interactions that exploits the phenomenon of surface plasmon resonance.2
There are a large number of different optical setups that are used in SPRi with their own advantages and shortcomings.3 Commonly, a polarized beam of light (as opposed to polychromatic light used in traditional SPR) is shone through a prism onto a 2D array of binding sites on thin films of gold.2,3 A Charge-Coupled Device (CCD) camera is used to capture high-resolution images of the binding area.2,3
In this technique, measurements are taken at a constant wavelength and angle.2,3 The change in reflected light intensity (which is proportional to the change in the refractive index of compounds bound to the metal film) is measured using images from the CCD camera.2,3 This setup could be used to theoretically study hundreds of binding sites simultaneously.2,3 However, the inherent limitations of using camera images for measurements (such as picture resolution) typically results in lower sensitivity measurements compared to other SPR techniques.4
When should SPRi be used?
With Surface plasmon resonance imaging, a significant number of interactions can be studied simultaneously across a 2D array of binding sites. This higher throughput technique is a great choice when there are several dozen interactions to screen. Analytes also do not need to be delivered through different fluidic channels, SPRi is often used in whole-cell studies. However, due to the sensitivity limitations of surface plasmon resonance imaging, it is recommended that promising candidates from an SPRi experiment be analyzed further with a more sensitive and reliable technique. Using a traditional SPR system that measures the change in incidence angle could be used here. Alternatively, a system that exploits localized surface plasmon resonance (LSPR) is another option.
LSPR: What makes OpenSPR innovative
Localized surface plasmon resonance (LSPR) uses gold nanoparticles – as opposed to a thin film of gold – to exploit the phenomena of SPR. When broadband white light is shone on the gold nanoparticles, a strong resonance absorbance peak is produced in the visible spectrum of light. This absorbance peak is highly sensitive to the local refractive index of materials bound near the particles. In this setup, the change in wavelength of absorbance can be measured, as opposed to the angle of incidence. As a result, the prism can be taken out of the equation, leaving a simpler and more robust detection system.
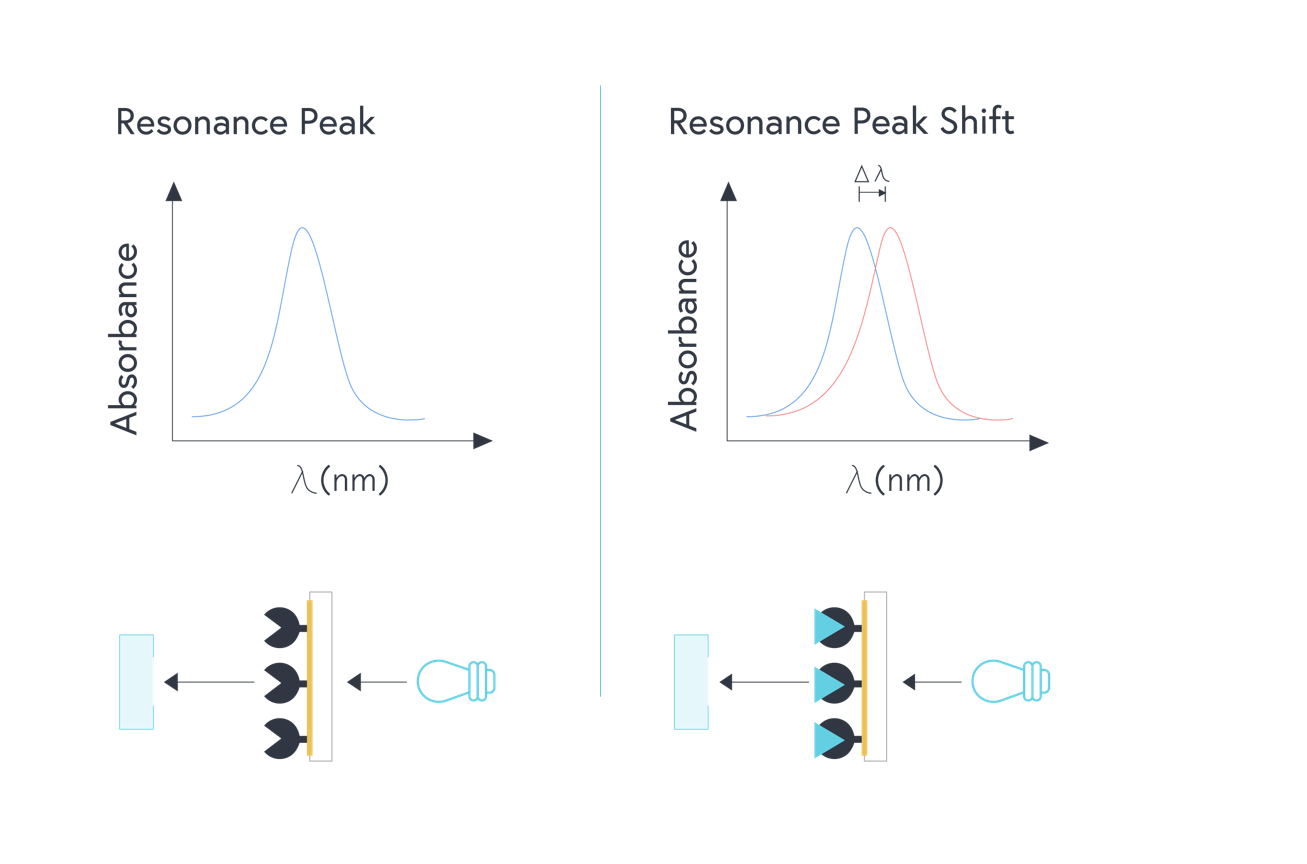
Figure 2: Illustration of LSPR optical system. Broadband white light shone onto a layer of gold nanoparticles with a ligand immobilized to the surface. As analytes bind to the surface, the resulting change in the refractive index changes the wavelength of absorbance that is measured.
As a result of this detection method, an SPR instrument that exploits LSPR can be designed at a much lower cost and requires significantly less maintenance. This enables the OpenSPRTM to be more affordable – costing nearly ten times less than leading competitors and with no annual service fees. The OpenSPRTM was also designed to be intuitive enough for a high school student to operate, making it an excellent choice for a lab that is new to SPR.
With the increased demand for high throughput techniques in biomedical research, surface plasmon resonance imaging is a great choice for measuring the binding kinetics of a large number of interactions. However, when it’s time to push drug candidates to the next stage of research, use a technique that gets the sensitive data you need.
References
- Rockberg, J., Nilvebrant, J. (2018). Epitope Mapping Protocols (3rd ed., Vol. 1785). Doi: 10.1007/978-1-4939-7841-0_1
- Nguyen, H.H., Park, J., Kang, S., Kim, M. (2019). Surface Plasmon Resonance: A Versatile Technique for Biosensor Applications. Sensors, 15(5), 10481-10510. Doi:10.3390/s150510481
- Wang, D., Loo, J.F.C., Chen, J., Yam, Y., Chen, S.C., He, H., Kong, S.K., Ho, H.P. (2019). Recent Advances in Surface Plasmon Resonance Imaging Sensors. Sensors, 19(1266), 1-26. Doi:10.3390/s19061266
- Laplatine, L. (2014). Spatial Resolution of Prism based Surface Plasmon Resonance Microscopy (Doctoral Dissertation). Université De Grenoble, Grenoble, France.