For decades, Green Fluorescent Protein (GFP) has been used as a novel cell and molecular biology tool with applications in imaging and reporting assays. Notably, expression and localization of proteins of interest can be tracked live in vivo using a GFP fusion tag. However, the limitation to using GFP fusions include the restriction of resolution in a variety of applications. For that reason, it is crucial to have GFP fusion alternatives to address the limitations inherent to this methodology.
In this new publication, Dr. Stasevich et al. used localized surface plasmon resonance technology, OpenSPR, in their latest discovery. The writing found in Nature Communications titled, “A genetically encoded probe for imaging nascent and mature HA-tagged proteins in vivo” uses binding kinetics data generated from OpenSPR to explore the feasibility of using an engineered high-affinity probe for use in live-cell imaging techniques.
[et_pb_section bb_built=”1″ admin_label=”section”][et_pb_row admin_label=”row” background_position=”top_left” background_repeat=”repeat” background_size=”initial”][et_pb_column type=”4_4″][et_pb_text admin_label=”Paragraph 1″ background_layout=”light” text_orientation=”justified” text_font_size=”16″ text_line_height=”1.9em” use_border_color=”off” custom_margin=”-25px|||” custom_padding=”||10px|” background_position=”top_left” background_repeat=”repeat” background_size=”initial” _builder_version=”3.0.96″]
About the Publication
[/et_pb_text][/et_pb_column][/et_pb_row][et_pb_row background_position=”top_left” background_repeat=”repeat” background_size=”initial”][et_pb_column type=”4_4″][et_pb_text admin_label=”Paragraph 2″ background_layout=”light” text_font_size=”16″ text_line_height=”1.9em” use_border_color=”off” custom_margin=”-20px|||” background_position=”top_left” background_repeat=”repeat” background_size=”initial” _builder_version=”3.0.96″ /][et_pb_text admin_label=”Paragraph 2″ background_layout=”light” text_font_size=”16″ text_line_height=”1.9em” use_border_color=”off” custom_margin=”-20px|||” background_position=”top_left” background_repeat=”repeat” background_size=”initial” _builder_version=”3.0.96″ header_line_height=”5px”]
GFP is notorious for long maturation times and does not allow for short-lived events such as transcription and translation to be observed; proteins are often degraded by the time GFP has fully matured. Moreover, post-translational modifications and conformation changes are challenging to resolve due to the long maturation time of GFP. However, antibody-based probes for use in live-cell imaging have been identified and may prove sufficient to address the limitations mentioned above.
In antibody-based methodologies for live-cell imaging, antigen-binding fragments (Fabs) and single-chain variable fragments (scFvs) are fused with mature fluorophores. This technique adequately addresses the previously described limitations with GFP (i.e. post-translational modification/conformational change resolution, increasing tracking-time, etc.). While Fabs are relatively straightforward to develop, they present unique challenges such as a limited ability to load into in vivo systems and can vary from batch to batch. In contrast, genetically encoded probes can be incorporated easily into living systems and drastically reduce variability among batches.
In this publication, the investigators engineered a hybrid scFv imaging probe termed the ‘HA frankenbody,’ targeting the HA epitope. Many in vivo techniques are carried out to confirm the robustness, specificity, and affinity of the probe, including Western blotting, immunostaining, colocalization and FRAP assays. Upon experiment conduction, the investigators found that the HA frankenbody probe can adequately resolve HA-tagged cytoplasmic, nuclear, membrane and mitochondrial proteins in a variety of cell types and conditions. Importantly, FRAP assays revealed that a majority of the engineered probe allows for minutes of live-cell tracking, indicating a high-affinity for its epitope. Surface Plasmon Resonance was used to quantify on/off rates, affinity, and confirm the overall efficacy of the engineered probe.
[et_pb_section bb_built=”1″ admin_label=”section”][et_pb_row admin_label=”row” background_position=”top_left” background_repeat=”repeat” background_size=”initial”][et_pb_column type=”4_4″][et_pb_text admin_label=”Paragraph 1″ background_layout=”light” text_orientation=”justified” text_font_size=”16″ text_line_height=”1.9em” use_border_color=”off” custom_margin=”-25px|||” custom_padding=”||10px|” background_position=”top_left” background_repeat=”repeat” background_size=”initial” _builder_version=”3.0.96″]
Why was OpenSPR instrumental for this research?
[/et_pb_text][/et_pb_column][/et_pb_row][et_pb_row background_position=”top_left” background_repeat=”repeat” background_size=”initial”][et_pb_column type=”4_4″][et_pb_text admin_label=”Paragraph 2″ background_layout=”light” text_font_size=”16″ text_line_height=”1.9em” use_border_color=”off” custom_margin=”-20px|||” background_position=”top_left” background_repeat=”repeat” background_size=”initial” _builder_version=”3.0.96″ /][et_pb_text admin_label=”Paragraph 2″ background_layout=”light” text_font_size=”16″ text_line_height=”1.9em” use_border_color=”off” custom_margin=”-20px|||” background_position=”top_left” background_repeat=”repeat” background_size=”initial” _builder_version=”3.0.96″ header_line_height=”5px”]
Surface plasmon resonance (SPR) was used to directly measure binding constants between purified HA epitope tag and HA frankenbody. Biotinylated HA peptide is immobilized on a streptavidin sensor chip, and HA frankenbody is diluted in a PBS running buffer. Two concentrations (100 nM and 30 nM) of HA frankenbody are injected and allowed 5 minutes of interaction time per injection. The assay is timed for ten minutes to measure dissociation and a 1:1 binding model is employed using a TraceDrawer to obtain kinetic constants. Notably, the KD measured by the OpenSPR (14.7 ± 7.4 nM) is consistent with results obtained from FRAP assays.
The investigators posture that high-affinity imaging probes translate to increased imaging resolution and in this respect, OpenSPR binding data allowed researchers to confirm the overall efficacy of their HA frankenbody probe as a novel imaging tool. By using OpenSPR, the researchers can get SPR data from their benchtop, accelerating their research and publishing their discovery faster.
[/et_pb_text][/et_pb_column][/et_pb_row][et_pb_row background_position=”top_left” background_repeat=”repeat” background_size=”initial”][et_pb_column type=”4_4″][et_pb_text admin_label=”Paragraph 2″ background_layout=”light” text_font_size=”16″ text_line_height=”1.9em” use_border_color=”off” custom_margin=”-20px|||” background_position=”top_left” background_repeat=”repeat” background_size=”initial” _builder_version=”3.0.96″ /][et_pb_text admin_label=”Paragraph 2″ background_layout=”light” text_font_size=”16″ text_line_height=”1.9em” use_border_color=”off” custom_margin=”-20px|||” background_position=”top_left” background_repeat=”repeat” background_size=”initial” _builder_version=”3.0.96″ header_line_height=”5px”]
Why is SPR critical for publications? How does OpenSPR help?
[/et_pb_text][/et_pb_column][/et_pb_row][et_pb_row background_position=”top_left” background_repeat=”repeat” background_size=”initial”][et_pb_column type=”4_4″][et_pb_text admin_label=”Paragraph 2″ background_layout=”light” text_font_size=”16″ text_line_height=”1.9em” use_border_color=”off” custom_margin=”-20px|||” background_position=”top_left” background_repeat=”repeat” background_size=”initial” _builder_version=”3.0.96″ /][et_pb_text admin_label=”Paragraph 2″ background_layout=”light” text_font_size=”16″ text_line_height=”1.9em” use_border_color=”off” custom_margin=”-20px|||” background_position=”top_left” background_repeat=”repeat” background_size=”initial” _builder_version=”3.0.96″ header_line_height=”5px”]
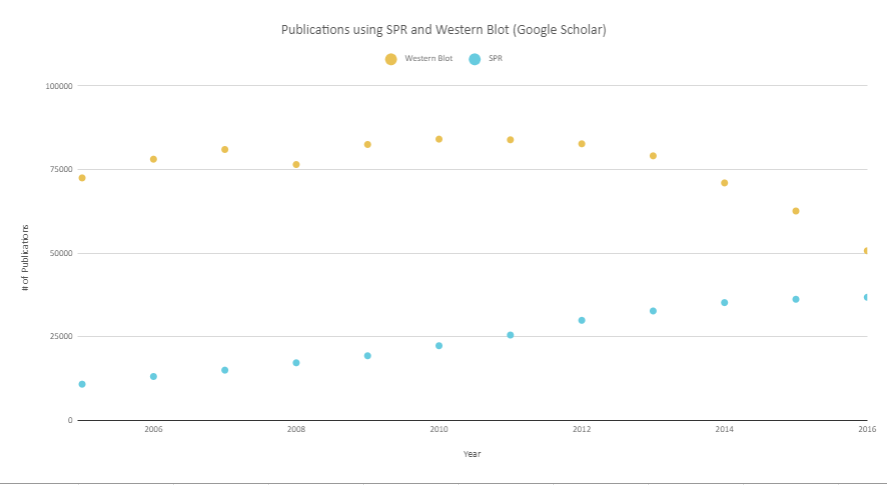
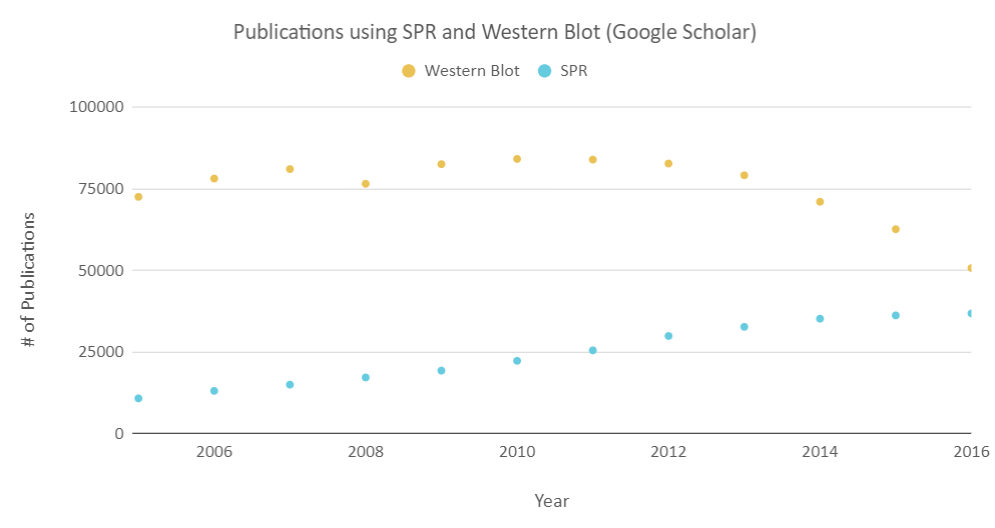
Surface Plasmon Resonance is a label-free technology which allows researchers to analyze binding between two biomolecules quantitatively. SPR technology will enable us to determine the kon, koff and KD of interactions, providing more in-depth insight into binding events compared to other techniques that only give endpoint measurements, such as pull-down assays. SPR is necessary not only for publications but for the advancement of many fields of medicine and medical research as can be seen below with the significant increase in papers that rely on SPR data.
[et_pb_section bb_built=”1″ admin_label=”section”][et_pb_row admin_label=”row” background_position=”top_left” background_repeat=”repeat” background_size=”initial”][et_pb_column type=”4_4″][et_pb_text admin_label=”Paragraph 1″ background_layout=”light” text_orientation=”justified” text_font_size=”16″ text_line_height=”1.9em” use_border_color=”off” custom_margin=”-25px|||” custom_padding=”||10px|” background_position=”top_left” background_repeat=”repeat” background_size=”initial” _builder_version=”3.0.96″]
[/et_pb_text][/et_pb_column][/et_pb_row][et_pb_row background_position=”top_left” background_repeat=”repeat” background_size=”initial”][et_pb_column type=”4_4″][et_pb_text admin_label=”Paragraph 2″ background_layout=”light” text_font_size=”16″ text_line_height=”1.9em” use_border_color=”off” custom_margin=”-20px|||” background_position=”top_left” background_repeat=”repeat” background_size=”initial” _builder_version=”3.0.96″ /][et_pb_text admin_label=”Paragraph 2″ background_layout=”light” text_font_size=”16″ text_line_height=”1.9em” use_border_color=”off” custom_margin=”-20px|||” background_position=”top_left” background_repeat=”repeat” background_size=”initial” _builder_version=”3.0.96″ header_line_height=”5px”]