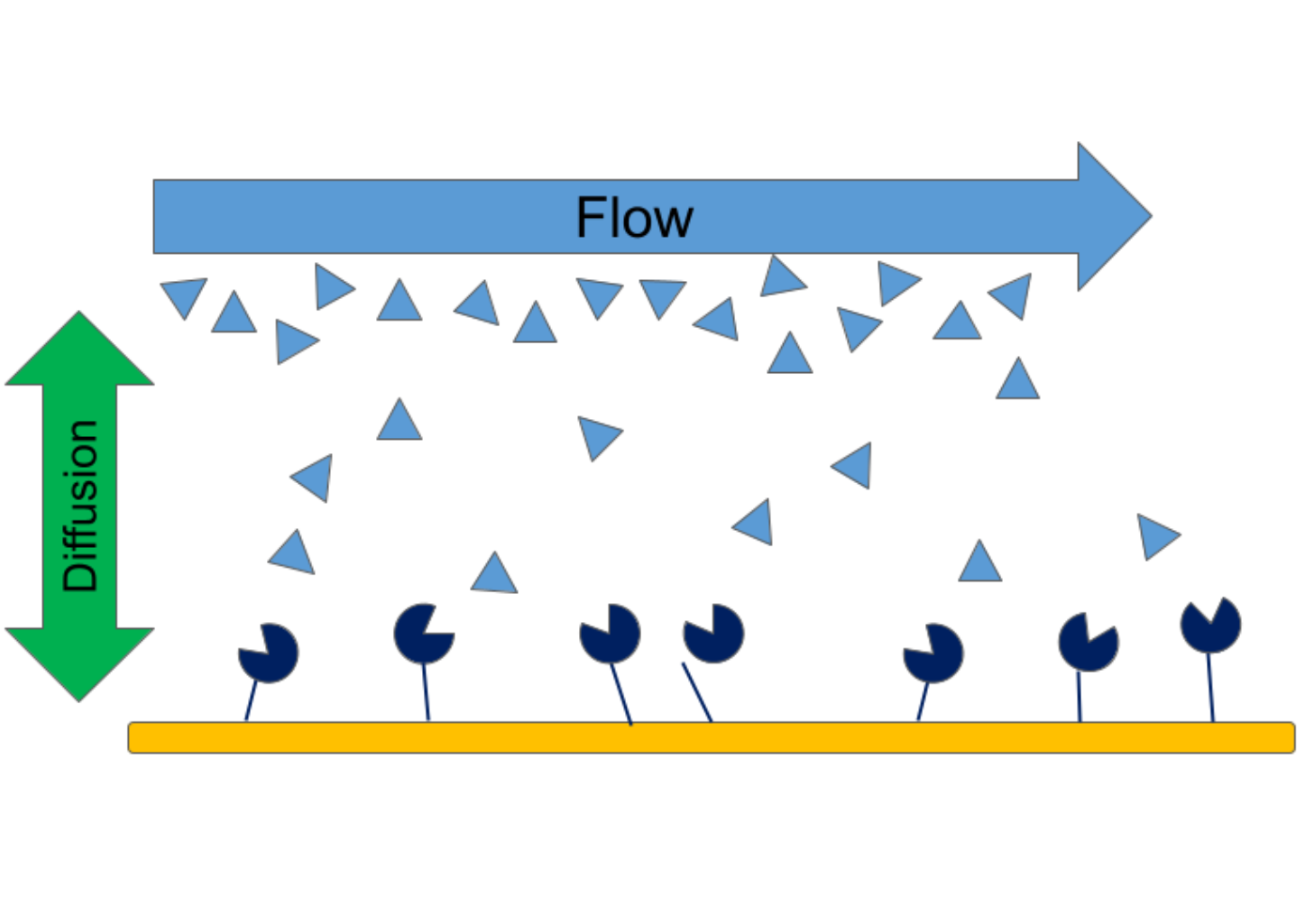
In surface plasmon resonance, the reactant in a solution must first diffuse from the bulk to the surface to interact with the immobilized ligand. If the diffusion rate is slower than the association rate, mass transfer effects can be observed in the data. Mass transfer limitations are most common for fast binding reactions, as diffusion limits the association rate. Here are three common ways one can limit mass transfer effects in kinetic data:
1) Higher flow rates
Increasing the rate of diffusion by increasing flow rate is an easy way to limit mass transport effects in surface plasmon resonance experiments. Increasing the flow rate will deliver the analyte to the immobilized ligand faster but won’t affect the kinetic data. A quick way to determine if you have mass transfer effects is to inject the analyte at a few different flow rates. If the Ka decreases at lower flow rates, the interaction is mass transport limited. The trade-off with increasing the flow rate too high (when using a small sample loop) is that the analyte might not have enough time in the association phase.
2) Lower surface density of ligand
The more ligand you have immobilized on your sensor, the more possible interaction sites for the analyte to bind. Reducing the amount of ligand immobilized to the sensor will help reduce diffusion-limited effects as less analyte needs to diffuse for the interaction to occur. This is especially key if the analyte is fast binding as you’ll want to reduce the ligand surface density to reduce diffusion-limited effects. The trade-off with lowering the surface density is a decrease in the Rmax (max signal at saturation), so the data will be noisier.
3) Mass transport corrected model
The best practice for accounting for diffusion limited effects in your surface plasmon resonance data is to use a fitting model that includes mass transport in the overall reaction equations. Most kinetic processing software include this model as an option when fitting data. The 1:1 Diffusion Corrected Model in the post-processing software TraceDrawer is the second model found in the drop down menu, so accounting for mass transport in your data is a simple click away! Using diffusion corrected models are good practice because even if you don’t have mass transfer limited effects in your data, your results won’t be affected by using the model. The only trade-off is that it could take a bit longer to fit the data.
Tip – If you’re curious whether you do have mass transport effects, you can fit your data to the 1:1 Langmuir model and the 1:1 Langmuir mass transport corrected model. If the ka is lower for the 1:1 Langmuir model, then your interaction is mass transport limited.
Learn more about mass transfer effects in surface plasmon resonance data:
http://www.sprpages.nl/data-fitting/models/mass-transfer
D.G. Myszka, T.A. Morton, M.L Doyle and I.M. Chaiken., “Kinetic analysis of a protein antigen-antibody interaction limited by mass transport on an optical biosensor.” Biophys Chem, vol. 64, pp. 127-137, 1997.
T.A. Morton, D.G. Myszka and I.M. Chaiken., “Interpreting complex binding kinetics from optical biosensors: a comparison of analysis by linearization, the integrated rate equation, and numerical integration.” Anal Biochem, vol. 227, pp.176-185, 1995.
Karlsson, H. Roos, L. Fagerstam and B. Persson., Methods: Companion to Methods In Enzymology, Academic Press.” vol. 6, pp. 99, 1994.
R.J. Fischer, M. Fivash, J. Casas-Finet, S. Bladen and K.L. McNitt.,Methods: Companion to Methods in Enzymology., vol. 6, pp. 121-133, 1994.