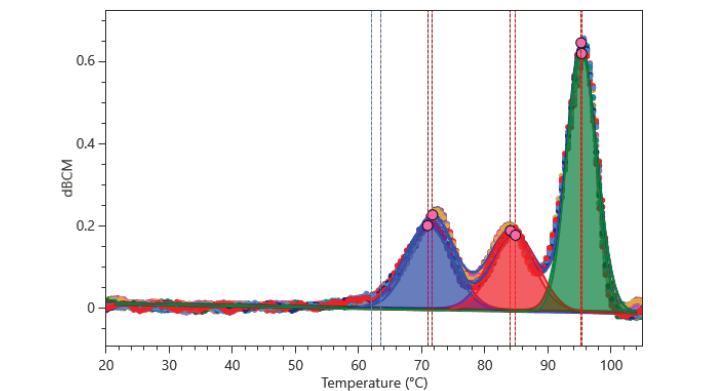
The development of biosimilar monoclonal antibodies requires rigorous demonstration of structural and functional equivalence to the reference product under a regulatory “totality of evidence” framework. In this study, we present an integrated, multi-dimensional biophysical characterization of Tocilizumab, a humanized IgG1 monoclonal antibody targeting the interleukin-6 receptor (IL-6R), and a biosimilar candidate. Higher order structure (HOS), thermal stability, and binding kinetics were systematically evaluated using a complementary suite of orthogonal techniques: Digital Surface Plasmon Resonance (Digital SPR™), differential scanning fluorimetry (SUPR-DSF™), and circular dichroism (CD) spectroscopy (Chirascan™ V100).