What is HDX-MS?
Hydrogen-Deuterium Exchange Mass Spectroscopy (HDX-MS) is a powerful technique for researchers to study protein interactions and conformational changes. The technique involves exposing proteins to heavy water (D2O), allowing deuterium to replace the hydrogen atoms in the protein backbone, with exposure times varying from a few seconds to hours. The rate at which this exchange happens in various regions of the protein is indicative of their level of exposure to the deuterium and provides useful information about the protein’s structure and dynamics.
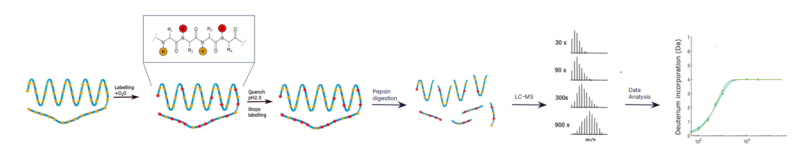
Deuterium uptake in disordered proteins and peptides, rapid small molecule binding and transient conformational changes occur within milliseconds. With a minimum labeling time of 30 seconds, traditional HDX is unable to capture these events, creating development blind spots.
Meet FastHDX
FastHDX brings hydrogen deuterium exchange into the millisecond timescale, enabling researchers to observe rapid structural dynamics that shape protein function and interaction. By capturing how proteins respond to different stimuli, like binding or environmental changes in real time, FastHDX provides deeper mechanistic insight into structure, stability, and conformational behavior.
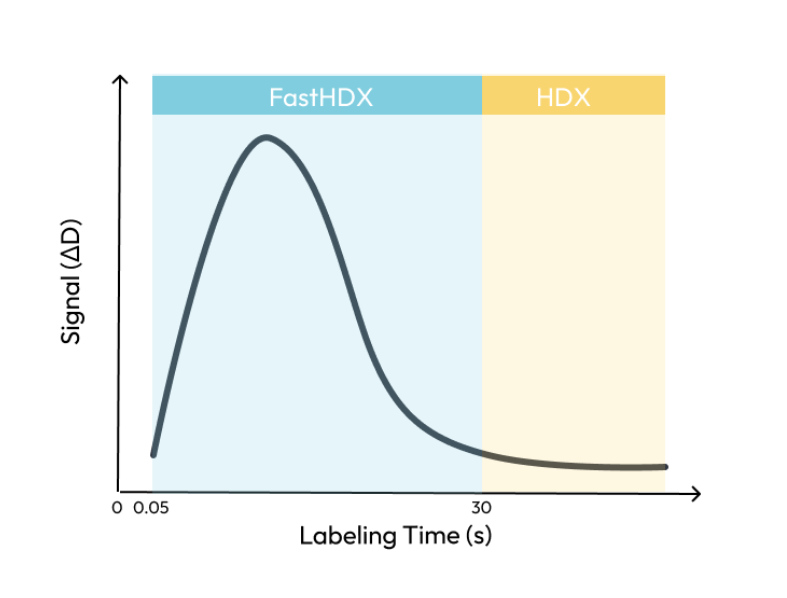
FastHDX opens new development avenues
FastHDX enables scientists to find and design therapies against previously inaccessible targets by revealing protein behaviour that conventional approaches miss. This innovative technology can be leveraged to find and design therapies for new targets through avenues like:
Allostery
Map and understand transient, non-equilibrium states and transitions resulting from allosteric regulation.
While Glycogen Phosphorylase was the very first allosteric enzyme to be described, its structural regulation was not understood until recently, when FastHDX was used to quantitatively prove the decades old hypothesis that GlyP regulation is achieved through gating of the active site by (dis)order transitions1 As a proven therapeutic target for diabetes, cancers and neurodegenerative diseases 2, 3, 4, 5 a deeper structural understanding of glycogen phosphorylase may pave the way to better, more efficient therapies.
Small molecule binding
Identify and characterize weak allosteric binders that are missed by traditional systems, or require tedious NMR.
For example, Ascinimib was structurally developed over 2 years by Novartis using NMR because weak binding fragments could not be measured with HDX. FastHDX immediately identifies weak binders, condensing years-long development timelines into just weeks of testing.
Intrinsically Disordered Proteins (IDPs) & Intrinsically Disordered Regions (IDRs)
IDPs and IDRs have long been included in the category of “undruggable” targets – molecules which were considered impossible to treat with conventional approaches due to their continually changing shapes, making it impossible to identify potential binding pockets. With millisecond resolution, FastHDX enables structural measurements of these rapidly changing structures, allowing for the development of drugs capable of targeting them.
For example, α-Synuclein is a 140 amino acid intrinsically disordered protein in solution involved in neurotransmitter release. Obtaining structural information on the disordered protein is impossible with traditional HDX, making it difficult to develop therapies targeting it. FastHDX analysis of α-Synuclein yielded 100% coverage and peptide redundancy provided an average structural resolution of 1.4 amino acids.
Peptide mapping
With their smaller size and frequent lack of secondary structure peptides are much more exposed to solvents, and so take up deuterium much faster than larger proteins, making their structural mapping challenging with traditional HDX-MS 6. With millisecond resolution, the structure (or lack thereof) of peptides as they are conjugated or exposed to different solvents can be assessed, allowing for the development of new peptide and peptide-targeting therapeutics.
HDX-MS at millisecond resolution
FastHDX is a significant development in the field of HDX-MS, enabling researchers to expand areas of development. With an optimized workflow designed to minimize back exchange, millisecond labeling capability, and precise and robust control via quench flow technology, FastHDX is an HDX innovation that expands the view on molecular motions and alleviates development blind spots.
References:
- Kish, M.; Subramanian, S.; Smith, V.; Lethbridge, N.; Cole, L.; Vollmer, F.; Bond, N. J.; Phillips, J. J. Allosteric regulation of glycogen phosphorylase by order/disorder transition of the 250′ and 280s loops. Biochemistry 2023, 62, 1360–1368.
- Hoover, D. J.; Lefkowitz-Snow, S.; Burgess-Henry, J. L.; Martin, W. H.; Armento, S. J.; Stock, I. A.; McPherson, R. K.; Genereux, P. E.; Gibbs, E. M.; Treadway, J. L. Indole-2-carboxamide Inhibitors of Human Liver Glycogen Phosphorylase. J. Med. Chem. 1998, 41, 2934− 2938.
- Oikonomakos, N. G. Glycogen phosphorylase as a molecular target for type 2 diabetes therapy. Curr. Protein Pept. Sci. 2002, 3, 561− 586.
- Zois, C. E.; Harris, A. L. Glycogen metabolism has a key role in the cancer microenvironment and provides new targets for cancer therapy. J Mol Med (Berl) 2016, 94, 137−154.
- Brown, A. M.; Ransom, B. R. Astrocyte glycogen and brain energy metabolism. Glia 2007, 55, 1263−1271.
- Stofella, M.; Seetaloo, N.; St John, A. N.; Paci, E.; Phillips, J. J.; Sobott, F. Recalibrating protection factors using millisecond hydrogen/deuterium exchange mass spectrometry. Anal. Chem. 2025, 97, 2648–2657.



