Compliance, meet convenience – all in the Alto GxP Suite
The world’s most user-friendly SPR system can now support regulated workflows


Streamline development, enhance compliance
The features in the Alto GxP software expansion are specifically designed to support compliance with the FDA’s 21 CFR Part 11 guidelines.
Characterizing biomolecular interactions is essential throughout biologics discovery and development, and maintaining data integrity is critical at every step. The Alto Digital SPR system offers unmatched ease of use along with excellent precision and accuracy for binding kinetics measurements.
The GxP Suite further enhances the platform with specialized software and qualification services that introduce audit-ready controls and enhanced traceability. With these capabilities, Alto can support compliant characterization from early screening through late-stage drug R&D.
Control Platform Access
The GxP software expansion offers robust data protection and integrity with user access controls, password policies, and edit-protected data formats and storage. The software expansion also supports up to three configurable signature levels and reasons. Once signed, results are locked to prevent further modifications.
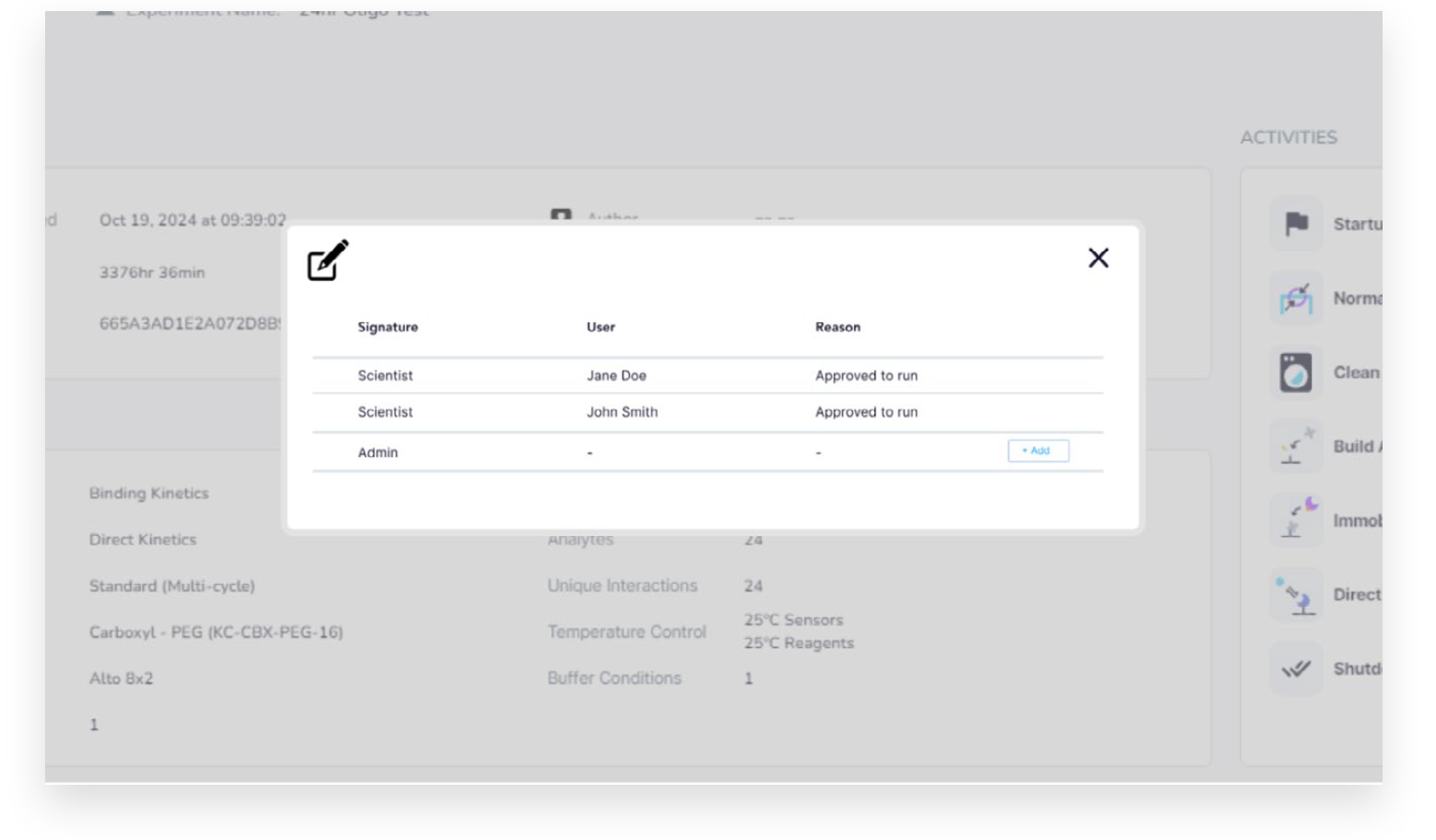
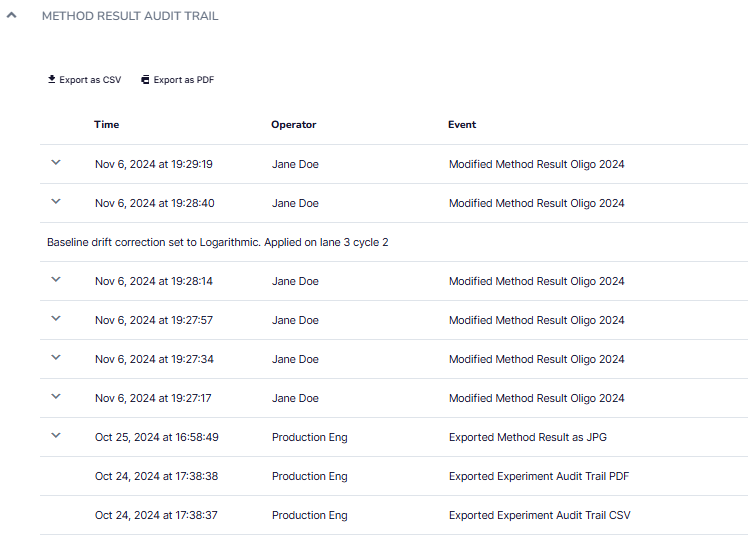
Create Comprehensive Audit Trails
The GxP software expansion maintains detailed audit trails to ensure full traceability and compliance. Every modification to methods and result files is recorded with timestamps and user information. Audit trails also track system events, including logins, diagnostics, and calibration tests, providing a complete history to support data integrity and regulatory requirements.
Ensure Your Alto Meets Specifications
Our comprehensive qualification services and kit provide confidence in reliable operation and adherence to stringent quality standards.
- On-site qualification service: A trained Nicoya representative will conduct installation qualification (IQ), preventive maintenance, and operational qualification (OQ).
- Qualification kit: Includes all required instructions and documentation for users to independently perform IQ, OQ, and performance qualification (PQ).
Resources
Analysis of antibody-antigen binding kinetics on Alto Digital SPR using mouse Fc-specific capture reagents
Overview Mouse Fc-specific capture reagents can be used to capture mouse antibodies or Fc-tagged proteins directionally. These capture reagents offer an optimal orientation for analyte…
Nicoya Alto CBX Cartridge Sensor Performance
With Alto, Nicoya’s Digital surface plasmon resonance™ (SPR) platform, the entirety of the fluidic control and sensing is done on single-use disposable cartridges. High accuracy…
Uncovering New Tools to Analyze Cytokine–Receptor Interactions: How Conigen Bioscience Used Nicoya’s Alto™ Digital SPR to Characterize Novel Dimeric Receptors
Presenters: Michael Piazza, PhD AVP, Applications Development Nicoya Lifesciences Alyssa Ball, PhD Professor, Assay Development Scientist Conigen Bioscience Overview Understanding cytokine-receptor interactions is critical for…

Stay compliant with Alto
Learn how the GxP suite can support your regulated workflows