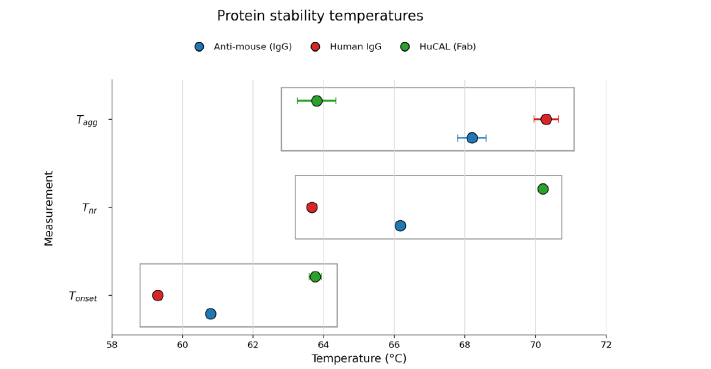
The development of biotherapeutics requires rigorous evaluation of both conformational and colloidal stability. In this application note, we introduce a new temperature cycling capability for the SUPR-DSF™ intrinsic differential scanning fluorimetry (DSF) platform, known in the literature as Modulated Scanning Fluorimetry (MSF). MSF enables direct assessment of protein folding reversibility, which correlates strongly with aggregation onset temperatures (Tagg) typically measured by Dynamic Light Scattering (DLS). Using representative protein systems, we demonstrate how MSF-derived insights complement traditional thermal stability (Tm) measurements. These results are obtained within a single experiment on the same instrument, while preserving the high throughput, low consumable cost, and minimal sample requirements of SUPR-DSF.